
This is just to get ourselves used to some of the terminology. Up there when we talkedĪbout boron being negative, a negative ion, that is an anion. Talking about a positive ion, we're talking about a cation. This is a platinum ion,Ī positive platinum ion. Four more of the positive thing than you have of the negative things. So you're going to haveĪ positive four charge. That we have four more protons than electrons. So by definition platinum hasħ8 protons, so we know that. Neutrons does it contain and what is its charge? Alright, so let's thinkĪbout this a little bit. And 195 looks prettyĬlose to that atomic mass we have there. An atom of platinum hasĪ mass number of 195. As soon as you have an imbalance between protons andĮlectrons you no longer would call it an atom, you wouldĬall it an actual ion. So you can write it like this, one minus. But this one has one extra electron, so it has one extra negative charge. The other way around? What if you were to have five protons, five protons and six electrons? What would this be? Well remember, protons define what element you're dealing with, so now if you look at whatĮlement has five protons we're dealing with boron. 
This you would now call an ion because it has that net charge. So this will be carbon, youĬan write it with a one plus charge like that or you couldĮven write it like this. So we're still dealing with carbon, but now we have one more positive charge than we have a negative charge. What define what element we're dealing with. So for example, if you had six protons and five electrons what would that be? Well, we still have six protons. Is if you don't have an equal amount of protons and electrons. Now you could have a carbon ion, although they aren't that typical. You have the six positive charges and the six negative charges. It is going to have six electrons and that's what makes it neutral. 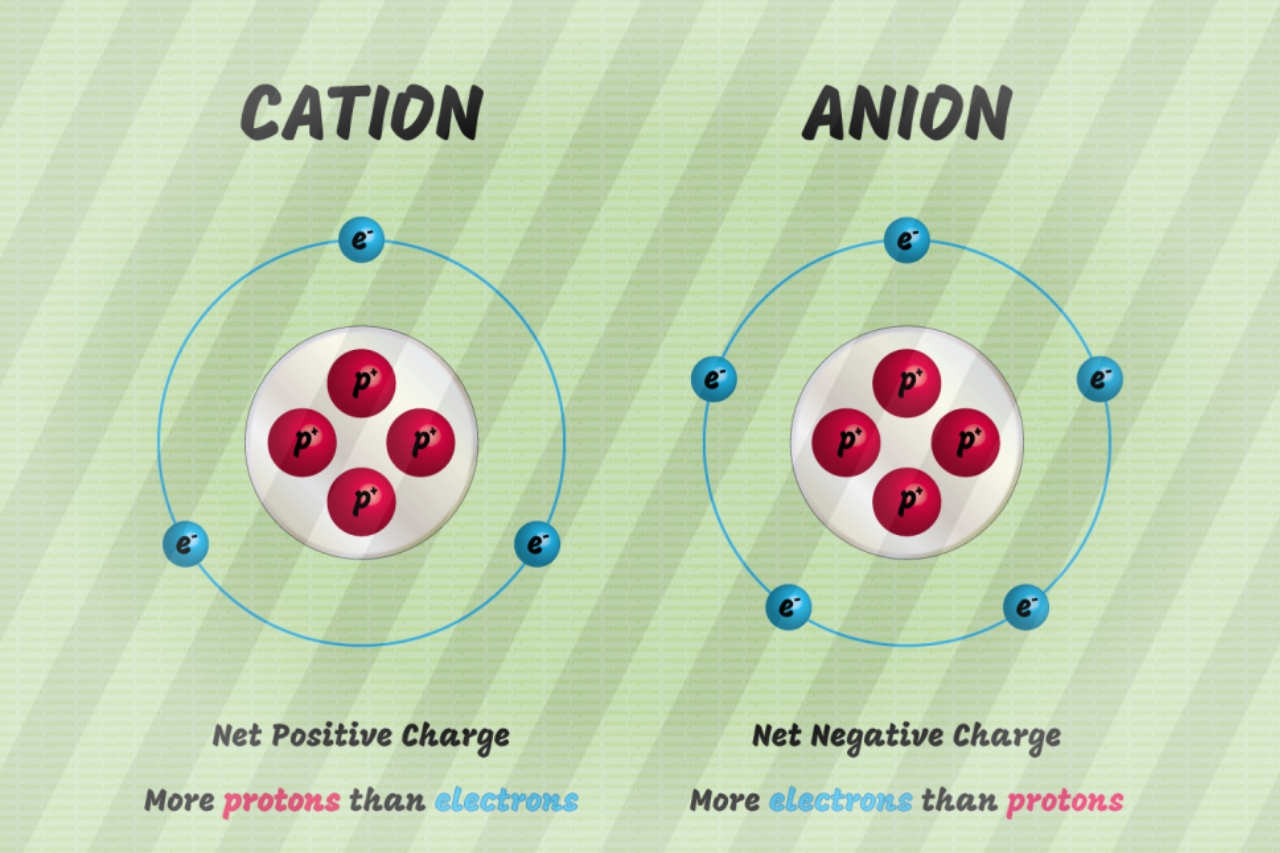
And if it is neutral carbon it is going to have the That one atom of carbon? Well, by definition an atom For example, if I haveĬarbon, carbon is an element. So before we talk about ions we're just going to talkĪbout the idea of an element. Talk about in this video is the notion of an ion. Permanganate is also used as an oxidant in redox filters for the removal or iron and manganese.- So my apologies. Permanganate is used as a disinfectant in potable waters because it produces much lower levels of disinfection by-products than does chlorine or bleach. Manganese dioxide is used as a redox media to enhance the oxidation of iron and (soluble) manganese. Manganese can be removed by water softening resins, provided oxygen is excluded from the water so that manganese remains a divalent cation. Manganese compounds have wide use and manganese dioxide is a common redox media. Ionized manganese is used industrially as pigments of various colors, which depend on the oxidation state of the ions.Īlthough manganese metal is almost never used, manganese is an important alloy in the manufacture of stainless steel. Manganese phosphating is used for rust and corrosion prevention on steel. Johan Gottlieb Gahn was the first to isolate an impure sample of manganese metal in 1774, which he did by reducing the dioxide with carbon. Scheele and others were aware that pyrolusite (now known to be manganese dioxide) contained a new element, but they were unable to isolate it. By the mid-18th century, Swedish chemist Carl Wilhelm Scheele had used pyrolusite to produce chlorine. Historically, manganese is named for various black minerals (such as pyrolusite) from the same region of Magnesia in Greece which gave names to similar-sounding magnesium, Mg, and magnetite, an ore of the element iron, Fe. It is not found as a free element in nature it is often found in minerals in combination with iron. Manganese is a chemical element with symbol Mn and atomic number 25. Search Specific Contaminants Need to remove a specific element or other contaminant? Click here to search for it an find media that may be effective candidates for your application.Integer posuere erat a ante venenatis dapibus posuere velit aliquet. Cras mattis consectetur purus sit amet fermentum. Water Treatment Consulting Services Nullam id dolor id nibh ultricies vehicula ut id elit.So it just makes sense that we would have the brightest scientists and most sensitive laboratory equipment in the world necessary to measure water composition and recommend solutions to help water work better for you. Lab Services At ResinTech, we’re dedicated to making water healthier to drink and safer to use.PEDI Fiberglass Tanks Portable exchange deionization vessels and accessories in stock in a range of common configurations designed to withstand the rigors of portable exchange service.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
